For patients who suffer from carcinoid syndrome – with symptoms such as diarrhea, frequent bowel movements, abdominal pain, and flushing – that is not adequately controlled by their current somatostatin analog therapy, there is an opportunity to participate in a global Phase III clinical trial of telotristat etiprate, an investigational oral treatment that inhibits the key enzyme responsible for the production of serotonin, tryptophan hydroxylase (TPH). This unique approach reduces the serotonin produced by functioning carcinoid neuroendocrine tumors (NETs) and helps to control the debilitating effects of carcinoid syndrome.
 The Phase III clinical trial of Lexicon Pharmaceutical’s telotristat etiprate (LX1606), also known as TELESTAR is now recruiting patients in over 50 study locations in the United States, Canada, Europe, and Australia. For additional information regarding this currently recruiting study or to see if you may qualify, visit www.telotristat-telestar.com.
The Phase III clinical trial of Lexicon Pharmaceutical’s telotristat etiprate (LX1606), also known as TELESTAR is now recruiting patients in over 50 study locations in the United States, Canada, Europe, and Australia. For additional information regarding this currently recruiting study or to see if you may qualify, visit www.telotristat-telestar.com.
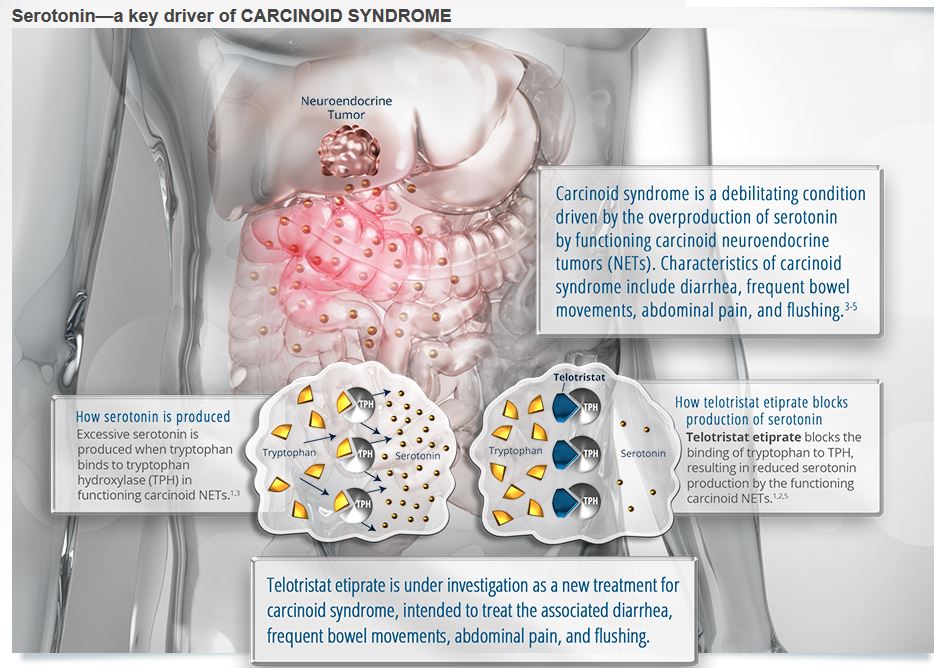
![]() Serotonin is a key driver of carcinoid syndrome. In the illustration below, see how serotonin is produced and how telotristat etiprate blocks serotonin production.
Serotonin is a key driver of carcinoid syndrome. In the illustration below, see how serotonin is produced and how telotristat etiprate blocks serotonin production.
Carcinoid syndrome is a chronic condition caused by metastatic neuroendocrine tumors that usually originate from the intestinal tract. This condition is caused by release into the circulation of excessive amounts of serotonin and other hormonal substances from some carcinoid tumors. Symptoms may include flushing of the face, diarrhea, bronchial spasms (wheezing), rapid pulse, and sudden blood pressure changes (usually a fall) along with other symptoms of heart failure. Carcinoid syndrome is often mistaken for common diseases including irritable bowel syndrome, rosacea, asthma, or menopause.
Telotristat etiprate reduces the ability of functioning carcinoid neuroendocrine tumors to produce serotonin by inhibiting tryptophan hydroxylase.
Lexicon is dedicated to providing travel assistance to patients where permitted. For more information please call the study hotline toll-free at 1-855-897-0125 (US and Canada) or 800-3000-2030 (international; be sure to include your country’s exit code, such as +00 for the EU), or e-mail at clinicalinfo@lexpharma.com.
For more details about the clinical trial, including criteria for participation, please visit: http://clinicaltrials.gov/ct2/show/NCT01677910?term=Telestar&rank=1. For additional information about TELESTAR, please visit http://telotristat-telestar.com/index.php. CLICK HERE to print out information about TELESTAR.