Tag: FDA
Lexicon’s Carcinoid Syndrome Treatment Meets Primary Endpoint in Phase III Clinical Trial
Lexicon Pharmaceuticals Inc. has announced that the pivotal phase III clinical trial, TELESTAR, of its oral drug candidate telotristat etiprate met the primary endpoint in treating cancer patients with carcinoid syndrome that is not adequately controlled…
READ MORE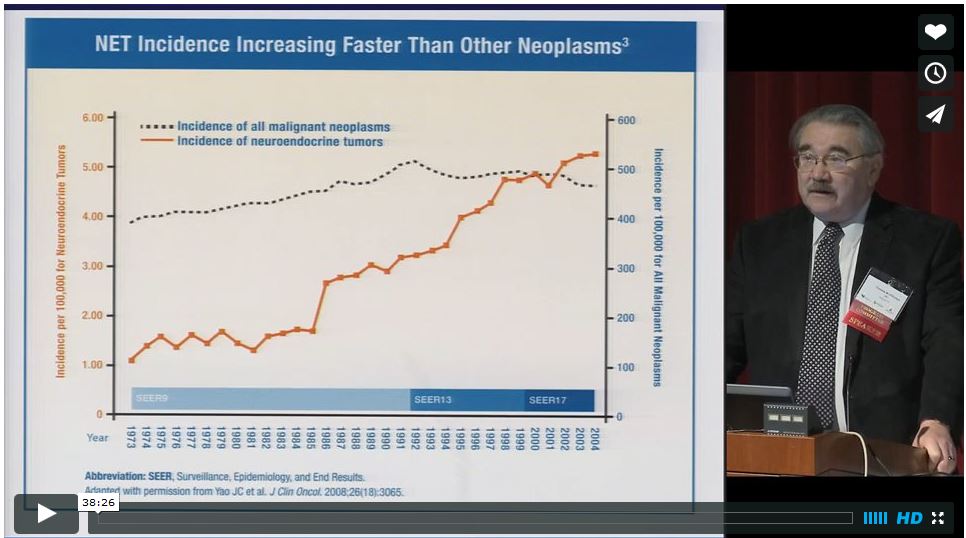
15 Videos from the 3rd Theranostics World Congress
The 3rd Theranostics World Congress featured a patient education day on Saturday, March 14, made possible with support from the Carcinoid Cancer Foundation and the Caring for Carcinoid Foundation. The NorCal CarciNET Community provided funding to…
READ MORE
Breaking News: US FDA Approves Lanreotide for Gastroenteropancreatic Neuroendocrine Tumors
December 16, 2014: The US Food and Drug Administration (FDA) today approved “lanreotide (Somatuline Depot Injection, Ipsen Pharma) for the treatment of patients with unresectable, well or moderately differentiated, locally advanced or metastatic…
READ MORELexicon Pharmaceuticals, Inc. Continues Recruiting for Phase 3 Studies in Carcinoid Syndrome
Many patients face the challenge of controlling carcinoid syndrome which can persist even while being treated with somatostatin analog (SSA) therapy. For patients on SSA therapy who continue to experience conditions such as diarrhea, frequent bowel…
READ MORE
10 Highlights of the Year 2013 for the Carcinoid and Neuroendocrine Tumor Community
As the Carcinoid Cancer Foundation looks back on the year 2013, we reached out to physicians, patients, support group leaders, listserv managers, patient advocates, and business colleagues to see what they thought were among the highlights of the past…
READ MORELexicon Pharmaceuticals Granted Orphan Drug Designation by FDA for Carcinoid Syndrome Treatment
Lexicon Pharmaceuticals, Inc. has been granted Orphan Drug Designation for telotristat etiprate (LX1032) by the U.S. Food and Drug Administration (FDA). LX1032 is used to treat carcinoid syndrome, a chronic condition caused by neuroendocrine tumors,…
READ MORE
Pancreatic Neuroendocrine Tumors: A Rare Cancer
From your next door neighbor to Steve Jobs, co-founder of Apple who just announced that he is stepping down as CEO of the company, pancreatic neuroendocrine tumors (pNETs) affect less than 1 person in 100,000 in the United States. This rare cancer is often…
READ MORE
Up-to-the-Minute: News for the Carcinoid and Neuroendocrine Tumor Community
The first device that simultaneously performs PET and MRI scans, a new alpha-emitter-based therapy for NET cancer patients with progressive therapy-resistant tumors, the first–ever randomized virtual clinical trial, over 100 abstracts on carcinoid…
READ MORE
Breaking News: FDA Approves Pfizer’s Sutent for Pancreatic NET Cancer Patients
The US Food and Drug Administration today announced approval of Pfizer’s Sutent (sunitinib) for advanced pancreatic neuroendocrine tumor cancer patients with tumors that cannot be removed by surgery or that have spread (metastasized) to …
READ MORE
Breaking News: FDA Approves Pfizer’s Sutent for Pancreatic NET Cancer Patients
The US Food and Drug Administration today announced approval of Pfizer’s Sutent (sunitinib) for advanced pancreatic neuroendocrine tumor cancer patients with tumors that cannot be removed by surgery or that have spread (metastasized) to …
READ MORE


