As we begin a new year, the Carcinoid Cancer Foundation looks back on the highlights of 2015 and to a future that we hope will bring greater awareness of carcinoid and neuroendocrine cancers, earlier diagnosis for patients, new treatment options, and continued research that will lead to a cure for these rare diseases. We reached out to patients, physicians, support group leaders, patient advocates, listserv managers, and business colleagues to see what they thought were the highlights of 2015.
Extremely promising results from 3 practice-changing Phase 3 clinical studies were considered the top highlights of the year by a nearly unanimous response from the NET community. In alphabetical order by study name, the top 3 highlights were:
1) NETTER-1
This international, randomized clinical study evaluated the efficacy and safety of 177-Lu-Dotatate (Lutathera ®) compared to high dose (60 mg) Octreotide LAR in patients with inoperable, progressive, somatostatin receptor positive, midgut neuroendocrine tumors. The primary objective of the trial was the assessment of Progression-Free Survival (PFS). According to carcinoid/NET specialist Dr. Jonathan Strosberg, “The findings were, in my opinion, extraordinarily impressive, the median progression-free survival improved by nearly 80%, which is fairly unprecedented in oncologic studies. The finding is important because limited therapeutic options exist for such patients, who comprise 20% to 45% of neuroendocrine tumor cases.” Study results were presented by NET specialist Dr. Philippe Ruszniewski for the first time at the European Cancer Congress during the Presidential Session on September 27, 2015 and for the first time in North America by Dr. Strosberg at NANETS in November 2015.
Advanced Accelerator Applications, maker of the drug, says “Lu-Dotatate is the most advanced candidate in development of peptide receptor radionuclide therapy (PRRT), which targets tumors with radiolabelled somatostatin analog peptides.” In April 2015, the FDA granted a fast track designation to Lu-Dotatate for the treatment of inoperable progressive midgut NETs. This designation is meant to facilitate the development of novel therapies.
Read more at: http://www.onclive.com/conference-coverage/nanets-2015/lu-dotatate-shows-unprecedented-pfs-in-midgut-nets#sthash.QBWMztLp.dpuf and http://www.adacap.com/netter-1/
2) RADIANT-4
The results of this Phase 3 pivotal study showed that Afinitor® (everolimus) tablets reduced the risk of progression by 52% vs placebo in patients with advanced, progressive, nonfunctional neuroendocrine tumors of gastrointestinal (GI) or lung origin. NET specialist Dr. James C. Yao, lead investigator, presented the results on behalf of the RADIANT-4 Study Group during the Presidential Session at the European Cancer Congress on September 27, 2015 and at the annual NANETS Symposium in November. According to Dr. Yao, “RADIANT-4 showed everolimus is the first targeted agent to show robust anti-tumor activity, with acceptable tolerability, against a broad spectrum of NETs, including those arising from the lung and GI tract. This is an area of a lot of unmet need. At this time, there is no drug that has ever demonstrated activity or been approved for lung neuroendocrine tumors, and that is roughly 25% to 30% of all neuroendocrine tumors out there. In progressive GI tumors, there are also limited options.”
http://https://www.youtube.com/watch?v=slWsUdNrhi0
Read more at: http://www.onclive.com/conference-coverage/nanets-2015/everolimus-shows-robust-activity-in-lung-gi-nets#sthash.kwbrFMuB.dpuf and https://www.novartis.com/news/media-releases/novartis-drug-afinitor%C2%AE-significantly-improves-progression-free-survival.
3) TELESTAR
 For patients with carcinoid syndrome who are no longer responding to the current standard of care, the result of this Phase 3 study was a decrease in daily bowel movements, some experiencing as high a percentage as 35% on a daily basis. According to NET specialist Dr. Matthew Kulke, principal investigator of the study, “These results clearly show telotristat etiprate represents a potentially promising new class of treatment for patients with carcinoid syndrome.” Lexicon Pharmaceuticals, based in Texas, received Fast Track designation and Orphan Drug status for telotristat etiprate from the U.S. Food and Drug Administration (FDA) in 2008 and 2012, respectively.
For patients with carcinoid syndrome who are no longer responding to the current standard of care, the result of this Phase 3 study was a decrease in daily bowel movements, some experiencing as high a percentage as 35% on a daily basis. According to NET specialist Dr. Matthew Kulke, principal investigator of the study, “These results clearly show telotristat etiprate represents a potentially promising new class of treatment for patients with carcinoid syndrome.” Lexicon Pharmaceuticals, based in Texas, received Fast Track designation and Orphan Drug status for telotristat etiprate from the U.S. Food and Drug Administration (FDA) in 2008 and 2012, respectively.
Read more at: http://www.onclive.com/conference-coverage/nanets-2015/novel-drug-relieves-carcinoid-syndrome-symptoms#sthash.I1jjkk8O.dpuf.
4) University of Iowa Receives First in Nation Grant to Study Neuroendocrine Tumors
Researchers at the University of Iowa Holden Comprehensive Cancer Center received the first-ever Specialized Programs of Research Excellence (SPORE) grant to study neuroendocrine tumors. The five-year, $10.67 million grant is the only SPORE grant funding research on neuroendocrine tumors. SPORE grants are funded through the National Cancer Institute (NCI), part of the National Institutes of Health. Carcinoid/NET specialist Dr. Sue O’Dorisio, professor of pediatrics in the Stead Family Department of Pediatrics and the UI Children’s Hospital, is the principal investigator on the SPORE grant. The grant will fund 4 major projects.
Read more at: http://now.uiowa.edu/2015/09/ui-researchers-receive-first-nation-grant-study-neuroendocrine-tumors.
5) Global NET Patient Survey
During 2015 results from the first-ever Global NET Patient Survey, developed through a collaborative effort by the International Neuroendocrine Cancer Alliance (INCA) and Novartis, were presented at medical/healthcare meetings in Australia, Europe, and North America. Completed by nearly 2,000 NET patients around the globe, the survey gathered information on the NET patient experience, including diagnosis, disease impact and management, quality of life, and knowledge and awareness levels. Key opinion leaders from the NET community in North America and Europe worked with representatives of INCA and Novartis to develop a master plan for presenting the survey results and many served as authors on abstracts/posters. The NET specialists who generously gave of their expertise and time are: Canada –Dr. Simron Singh; Europe –Dr. Dan Granberg, Dr. Kjell Oberg, Dr. Marianne Pavel, Dr. Philippe Ruszniewski; United States –Dr. Richard R.P. Warner, Dr. Edward Wolin, Dr. James C. Yao.
Posters were presented in 2015 at medical/healthcare conferences including the European Neuroendocrine Tumor Society (ENETS), the Academy of Managed Care Pharmacy, Oncology Nursing Society, the European Congress of Endocrinology, and the North American Neuroendocrine Tumor Society (NANETS). Dr. Edward Wolin, who gave a presentation at the largest annual American meeting of gastroenterologists, Digestive Diseases Week, said, “The gastroenterologists in the audience were amazed that approximately 50% of all NETs had been diagnosed with an incorrect GI diagnosis, most commonly irritable bowel syndrome or inflammatory bowel disease, although a large variety of other GI diagnoses were also entertained. The information provided by the Global NET Patient Survey is potentially practice-changing for the gastroenterologists in attendance, who will now think about the possibility of carcinoid earlier in a patient’s course, and attempt to make a diagnosis sooner with appropriate small bowel endoscopy and hormone measurements.”
Read more about the survey here: http://netcancerday.org/results-from-first-global-survey-of-neuroendocrine-tumor-net-patients/

6) NIH Study Finds Genetic Link in Gastrointestinal Carcinoid
The National Institutes of Health reported that “heredity accounts for up to 35 percent of small intestinal carcinoid,” based on an ongoing study of families with a history of the disease. Lead author on the study Dr. Stephen Wank noted that the study findings “suggest that people with a family history of the disease should be screened for it. We hope this research empowers thousands of at-risk people with a way to prevent these tumors from becoming a devastating disease.”
Read more from the NIH: http://www.nih.gov/news-events/news-releases/nih-study-finds-genetic-link-rare-intestinal-cancer. For those interested in the study, Natural History of Familial Carcinoid Tumor, click here: https://clinicaltrials.gov/ct2/show/NCT00646022?term=carcinoid+tumors&rank=2.
7) Warner Advocacy Award
 Congratulations to Josh Mailman upon being selected as the 2015 Warner Advocacy Award recipient! The award was established by Novartis Oncology in 2009 to commemorate the life and work of Monica Warner. Josh’s commitment to NET patient advocacy spans integrative oncology, nuclear medicine and molecular imaging, talking with patients, speaking at medical conferences around the world, and serving as President of the Northern California (NorCal) CarciNET Community. He also shares his technical expertise by redesigning websites and maintaining information systems for both the North American Neuroendocrine Tumor Society (NANETS) and the World Association of Radiopharmaceutical and Molecular Therapy (WARMTH).
Congratulations to Josh Mailman upon being selected as the 2015 Warner Advocacy Award recipient! The award was established by Novartis Oncology in 2009 to commemorate the life and work of Monica Warner. Josh’s commitment to NET patient advocacy spans integrative oncology, nuclear medicine and molecular imaging, talking with patients, speaking at medical conferences around the world, and serving as President of the Northern California (NorCal) CarciNET Community. He also shares his technical expertise by redesigning websites and maintaining information systems for both the North American Neuroendocrine Tumor Society (NANETS) and the World Association of Radiopharmaceutical and Molecular Therapy (WARMTH).
8) NET Cancer Day
2015 marked the 6th annual NET Cancer Day, an international day to raise awareness of neuroendocrine cancers. Each year the events presented on and around the day have grown in number and scope. From Australia to Europe and Asia to North America, events included conferences, walks and races, an international Twitter chat, distribution of awareness materials at medical facilities, and more. The International Neuroendocrine Cancer Alliance (INCA), sponsor of NET Cancer Day, presented the NET patient perspective at the November 10 meeting of the European Parliament, hosted by two recognized MEPs (elected officials to Parliament), who showed an exceptional level of engagement and support, thus giving the NET community a very strong platform upon which to build. In the United States, the Carcinoid Cancer Foundation, with the support of many NET patients, contacted every Governor in the country seeking proclamations, resolutions and letters of support for NET Cancer Awareness Day 2015. For the second consecutive year, a record forty-nine (49) states recognized the importance of early detection and proper treatment leading to improved outcomes and a better quality of life for carcinoid/NET (neuroendocrine tumor) cancer patients!! With support from Advanced Accelerator Applications, a zebra-striped filter was created for the NET community to use on their social media profiles and zebra-striped profile pictures were seen throughout the world.
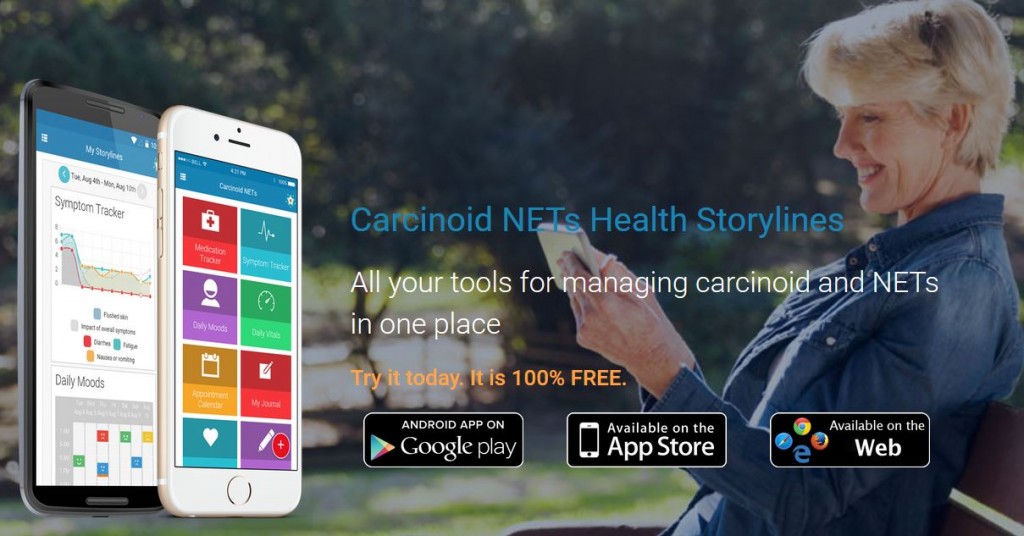
9) New Carcinoid NETs App
The Carcinoid Cancer Foundation, in partnership with Health Storylines/Self Care Catalysts, introduced a new app designed specifically for the carcinoid/NET community. Patients can record symptoms, nutrition, moods, medication, and more. The newest addition to the app is Healing Box Music, the soothing sounds of music composed by Giovanna Joyce Imbesi, while she was recovering from carcinoid surgery. App users can choose what to track to build their own health story. This enables them to learn more about their health and share more with their doctors.
The free app is available through the App store? and on Google Play™ only in the United States and Canada. The Web version is also available only for US and Canadian users at this time. Click here to read more and download the app: https://healthstorylines.com/blog/?page_id=476.
10) A Compilation
There are many additional noteworthy items and we list several of them here:
• Two NET specialists announced new locations of their practices:
Dr. Eric Liu along with medical oncologist Dr. Allen Cohn is heading up a new Neuroendocrine Tumor Center in Denver, Colorado.
Dr. Edward Wolin, effective in January, is relocating his practice to Montefiore Einstein Center for Cancer Care in Bronx, New York, where he will lead the Neuroendocrine Oncology team. http://www.montefiore.org/body.cfm?id=1735&action=detail&ref=6116
• After approval of lanreotide by the FDA in December 2014 for gastrointestinal-pancreatic neuroendocrine tumor patients, Ipsen began working closely with the NET community to determine the needs of patients and the advocacy community. Ipsen launched an updated website in 2015 about Somatuline Depot: https://www.somatulinedepot.com/gn/.
• The 3rd Theranostics World Congress on Ga-68 & PRRT, held in Maryland, included a free patient program on the final day of the Congress, made possible with sponsorship by the Carcinoid Cancer Foundation and the Neuroendocrine Tumor (NET) Research Foundation (formerly Caring for Carcinoid Foundation). The patients who attended the program had the opportunity to interact directly with representatives of the FDA.
• The annual North American Neuroendocrine Tumor Community (NANETS) symposium attracted nearly 350 healthcare professionals, a record number of attendees.
• A clinical trial, funded in part by the Neuroendocrine Tumor Research Foundation, has received the necessary regulatory approvals and is open to begin accruing patients. In particular, the clinical trial will enroll patients with carcinoid cancer for both treatment with the radionuclide Lutetium-177 and imaging with the radionuclide Gallium-68. Dr. Wolfgang Weber, and Diane Dr. Reidy-Lagunes, Memorial Sloan Kettering Cancer Center, are the lead researchers on this trial. The trial will employ a new approach to PRRT, the goal of which is to increase efficacy with fewer side effects than other PRRT trials of its kind. https://netrf.org/net-clinical-trial-opens-trial-to-assess-an-improved-approach-to-prrt-treatment/
• A new online carcinoid/neuroendocrine community premiered this year at Smart Patients, a program made possible by the Carcinoid Cancer Foundation in collaboration with Smart Patients. Join Smart Patients, ask a question, and support others who can learn from you: www.smartpatients.com/carcinoid.
 • Sadly, the NET community lost a staunch advocate and friend, Edda Gomez-Panzani, MD, who passed away on June 30, 2015. Dr. Gomez-Panzani, Vice President of Research & Scientific Affairs for Ipsen Biopharmaceuticals, was instrumental in gaining FDA approval for Somatuline Depot (lanreotide) for NETs patients in December 2014. Click here to read the Northern California (NorCal) CarciNET Community’s tribute to her.
• Sadly, the NET community lost a staunch advocate and friend, Edda Gomez-Panzani, MD, who passed away on June 30, 2015. Dr. Gomez-Panzani, Vice President of Research & Scientific Affairs for Ipsen Biopharmaceuticals, was instrumental in gaining FDA approval for Somatuline Depot (lanreotide) for NETs patients in December 2014. Click here to read the Northern California (NorCal) CarciNET Community’s tribute to her.
• TAPUR is ASCO’s first clinical trial to address the critical gaps in understanding of and access to targeted therapies. The American Society of Clinical Oncology is expanding the boundaries of precision medicine with the Targeted Agent and Profiling Utilization Registry (TAPUR) study. The non-randomized clinical study “aims to describe the performance (both safety and efficacy) of commercially available, targeted anticancer drugs prescribed for treatment of patients with advanced cancer that has a potentially actionable genomic variant.” Visit the TAPUR website here: http://www.tapur.org/.


